Home
Our Advantage
Direct collaboration, reliable services, and a wide range of expertise. See what makes us unique.
Quality In What We Do
We apply our experience in medical device design, rapid-prototyping, and test method development to provide novel solutions for state-of-the-art medical devices. We work with local medical device companies in Orange County and are expanding our services to other companies based on severely unmet needs.
Adherence to Regulation
The test method procedure developed complies with the FDA, ASTM, and ISO standards for intravascular devices to ensure the testing of clinically relevant specifications. The Force Detectors provided the equipment, set-up design, and the characterization, qualification, and validation procedures needed to implement the test method into production. There are no competitors for this test method because current methods are unable to properly test variably-shaped devices.

Rapid and Economical Response
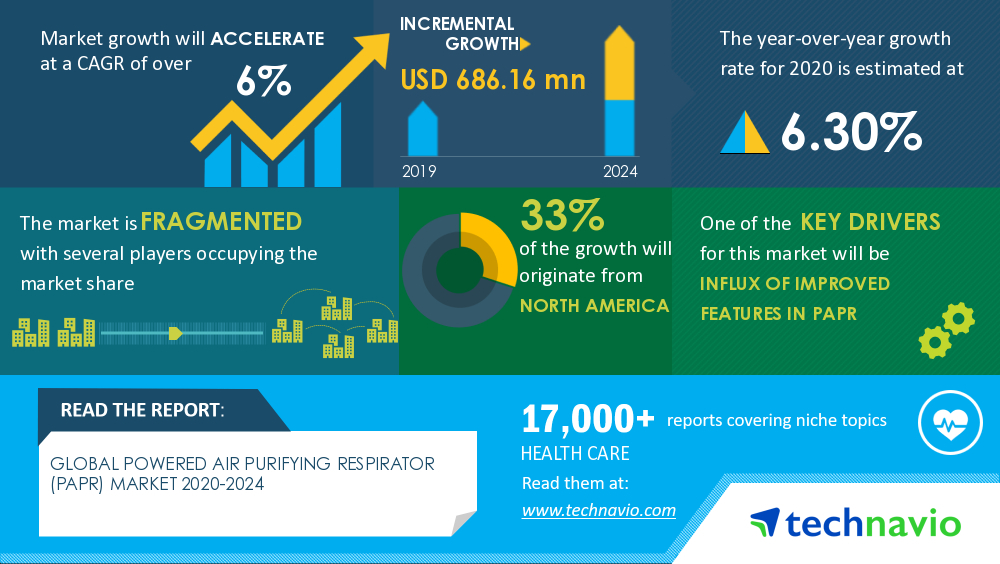
In response to the COVID-19 pandemic, the Force Detectors are working with other teams to develop and manufacture a controlled air purifying respirator (CAPR), a type of powered air purifying respirator (PAPR). The global market for PAPRs is expected to increase by over $686 million by 2024, with 33% of the growth originating from North America (Figure 2). To contribute to this growing market, the Force Detectors are developing a lighter CAPR for healthcare professionals.
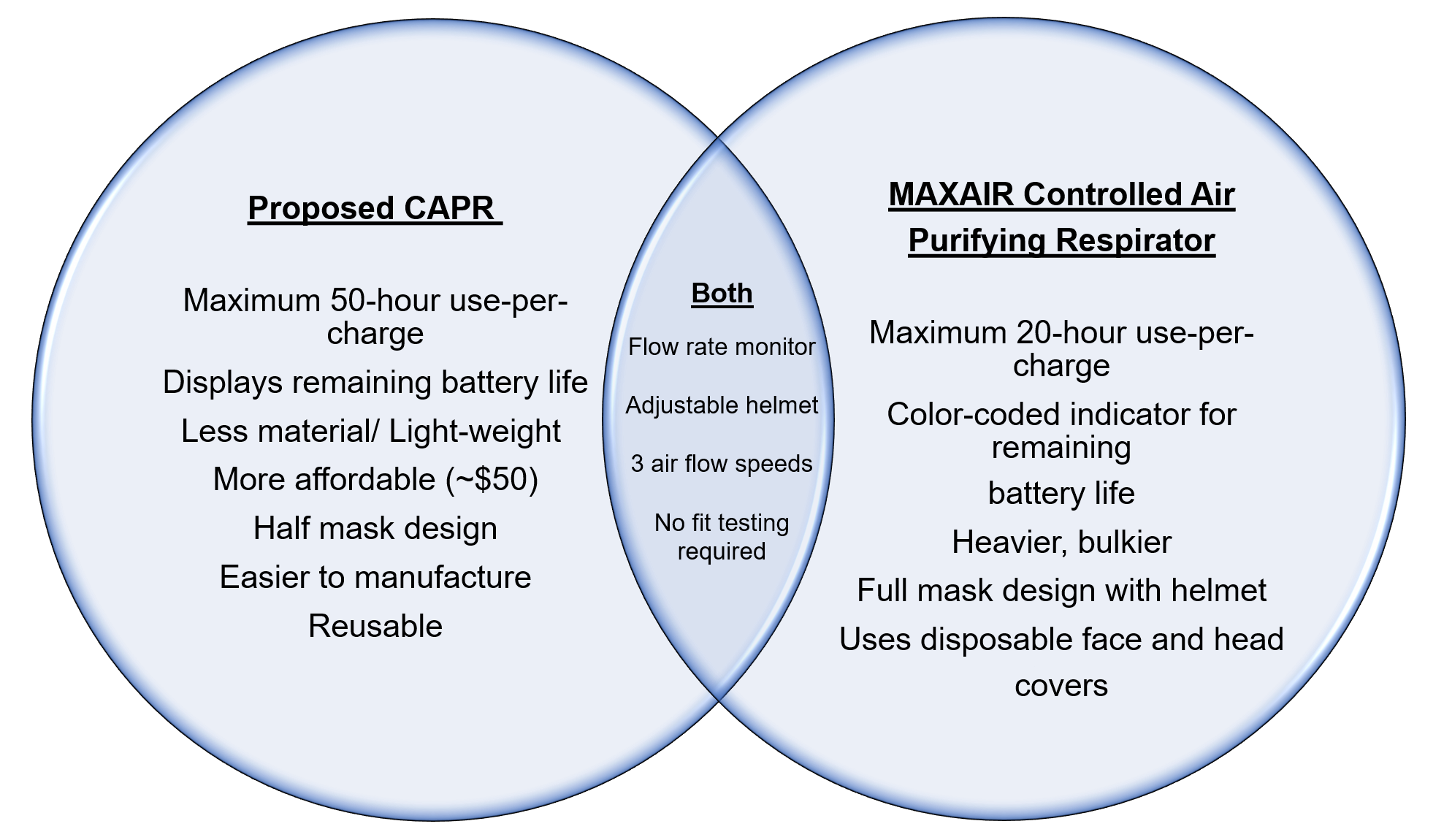
With the growing need for personal protective equipment due to the COVID-19 pandemic, the Force Detectors seek to design an affordable and easy to manufacture controlled air-purifying respirator (CAPR). CAPRs currently used in hospitals, including the MAXAIR® Controlled Air Purifying Respirator, are too expensive to afford for the increased demand and difficult to manufacture with the current supply chain disruptions. The proposed CAPR uses less material to provide a lighter system to protect our healthcare professionals and reusable components to make it more affordable than its competitors (Figure 3).